cell cycle
Cellular reproduction involves the proliferation of cells by replication of genetic material, followed by:
a) Binary fission in prokaryotes and plastids derived from prokaryotes (mitochondria , chloroplasts). In binary fission, which is a form of asexual reproduction, a cell divides transversely into two daughter cells after replication.
b) Budding (yeast, spores, Hydra) is a form of asexual reproduction in which a new organism buds from a cell protuberance in a form of unequal fission.
c) Mitotic division of somatic cells in which DNA is duplicated prior to fission into cells with an equivalent chromosomal complement to that of the parent cells.
c) Meiotic division of germ cells (sexual reproduction) in which fission generates daughter cells with half the chromosomal complement of the parent cells.
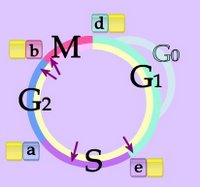 Control of the Cell Cycle (animation):
Control of the Cell Cycle (animation):
Right - Cell Cycle - click to enlarge image. Phases G1 and G0, S, G2, M. Checkpoints - purple arrows at G1-S transition, during S phase, G2-M transition, anaphase of mitosis. CDKs (yellow) are modulated by association with a series of fluctuating cyclins (d, e, a, b.) The cycle is divided into non-mitotic interphase (beige) with G1 or G0, S, and G2 phases followed by mitosis (pink).
Eukaryotic cells alternate genome doubling (S-phase, synthesis) with genome splitting (mitosis, M-phase) to generate daughter cells with an identical chromosomal complement. The cell cycle consists of a signal-controlled sequence of physiological states G1 → S → G2 → M, where non-mitotic phases are termed 'interphase' (beige) and G represents 'gap'. Quiescent cells are said to be in phase G0, in which they are not participating in the cell cycle, but are metabolically active.
In a normal resting cell, intracellular signaling proteins and genes remain inactive unless activated by extracellular growth factors. When the normal resting cell is stimulated by an extracellular growth factor, signaling proteins and genes are activated and the cell proliferates.
More than one hundred genes are specifically involved in cell cycle control, these are the so called CDC-genes (cell division cycle genes). One of these genes, designated CDC28 in Saccharomyces cerevisiae or CDK1 (cyclin dependent kinase 1) in humans, controls the first step in the progression through the G1-phase of the cell cycle, and is therefore also called "start". This gene encodes a protein member of the cyclin dependent kinase family (CDKs). Half a dozen different CDK molecules have been found in humans. Cyclins are proteins formed and degraded during each cell cycle. Cyclins are so-named because their levels vary periodically during the cell cycle. Cyclins bind to the CDK molecules, thereby regulating the CDK activity and selecting the proteins to be phosphorylated. Periodic protein degradation is an important general control mechanism of the cell cycle.
Cyclins were conserved during evolution. Around ten different cyclins have been found in humans. The levels of CDK-molecules are constant during the cell cycle, but their activities vary because of the regulatory function of the cyclins. CDK and cyclin together drive the cell from one cell cycle phase to the next. Levels of cyclins D (G1), E and A (S), and B and A (mitotic) fluctuate during the cell cycle (d,e,a,b) , and binding of appropriate cyclins to the cyclin-dependent kinases (CDKs) stimulating phosphorylation and activation.
The cell cycle is a highly coordinated process that is controlled at multiple checkpoints (purple arrows) along the pathway – the G1/S and G2/M transitions of interphase as well as anaphase of mitosis. These checkpoints are critical in preserving the fidelity of the genome. Alterations in the genes that control checkpoint processes are linked to a number of human malignancies including colon, breast, lung, kidney, brain and skin cancers.
During resting conditions, Rb sequesters the E2F transcription factor within the cytosol. Phosphorylation of Rb enables dissociation of the Rb-E2F complex, which allows E2F to translocate to the nucleus, where E2F binds to the promoter region of early genes that prime the cell to progress into the S-phase of the cycle. These early genes include c-myc, cyclin A/E, and CDK1(cdc2).
Progression through the cell cycle begins (G1 → S) with the commitment to undergo cellular division, which is initiated through the activation of the cyclin D-CDK4/6 complex and the subsequent phosphorylation of the Rb-E2F complex. Activity of the cyclin D-CDK4/6 complex is regulated through binding to its respective cyclin (4/6) counterpart, phosphorylation and dephosphorylation events, and the expression of a number of CDK inhibitors (CDKI’s), which . include p15 INK4, p16 INK4, p21 Cip-1 and p27 Kip-1. Progression through G1 is further facilitated by the cyclin E-CDK2 complex.
G1 → S
Rising level of G1-cyclins (D) bind to a G1 Cdk (Cdk 4), signaling the cell to prepare the chromosomes for replication.
DNA-damage checkpoints monitor DNA damage before the cell enters S phase (G1 checkpoint),
during S phase, and after DNA replication (G2 checkpoint). Increased levels of CDK-molecules and cyclins are sometimes found in human cancers. CDK-molecules and cyclins collaborate with the products of tumor suppressor genes, such as TP53 and Rb, during the cell cycle. The p53 protein senses DNA damage and can halt progression of the cell cycle in G1. Both copies of the p53 gene must be mutated for cycle arrest to fail, so mutations in p53 are recessive and p53 qualifies as a tumor suppressor gene. The protein generated by the p53 gene acts as a signal for apoptotic cell death when DNA damage is too extensive for repair mechanisms.
S → G2
Rising level of S-phase promoting factor (SPF) — which includes cyclin A bound to Cdk2 — enter the nucleus and prepare the cell to duplicate its DNA and its centrosomes. As DNA replication continues, cyclin E is destroyed, and the level of mitotic cyclins begins to rise (in G2).
In the S-phase of the cycle, the DNA is unwound and replicated. If DNA is damaged, the fate of the cell is determined by the extent of damage. If the damage is repairable, progression through the cycle will be halted in a p53-dependent matter through p21 Cip-1, allowing sufficient time to repair the damage.
Cells monitor the presence of the Okazaki fragments on the lagging strand during DNA replication, and cells do not proceed in the cell cycle until these have disappeared.
Entry into the M-phase of the cell cycle is regulated by the cyclin B-CDK1 complex. CDK1 activity is subject to multiple levels of control in an analogous fashion to the other cyclin-dependent kinases.
G2 → M
M-phase promoting factor (a complex of mitotic cyclins with the M-phase Cdk) initiates metaphase assembly of the mitotic spindle, breakdown of the nuclear envelope, condensation of chromosomes.
Spindle checkpoints monitor any failure of spindle fibers to attach to kinetochores and arrest the cell in metaphase until all the kinetochores are attached correctly (M checkpoint), detect improper alignment of the spindle itself, block cytokinesis, and trigger apoptosis if the damage is irreparable.
M-phase promoting factor activates the anaphase-promoting complex (APC/C, cyclosome) which allows the sister chromatids assembled at the metaphase plate to separate and move to the poles during telophase, completing mitosis. The APC/C destroys cyclin B by attaching it to the protein ubiquitin, thus targetting it for destruction by proteasomes (hydrolysis by proteases). APC/C also turns on synthesis of G1 cyclin for entry into the next turn of the cycle, degrades geminin, a protein that has retained the freshly-synthesized DNA in S phase to prevent if from being re-replicated before mitosis.
This is but one mechanism by which the cell ensures that every portion of its genome is copied once — and only once — during S phase. Some cells deliberately cut the cell cycle short allowing repeated S phases without completing mitosis and/or cytokinesis (endoreplication).
In a cancer cell, mutation in a proto-oncogene that encodes an intracellular signaling protein (normally activated only by extracellular growth factors) creates an oncogene. The oncogene encodes an altered form of the signaling protein that behaves as though activated despite the absence of growth factor binding. That is, the malignant cell has escaped normal gene regulation and cell cycle control mechanisms and exhibits unchecked proliferation.
Tables Regulatory Proteins Sequences Cell signaling Cell Adhesion Molecules Second Messengers Immune Cytokines Malignant Transformation Oncogenes Proto-oncogenes
Meiosis is controlled by similar factors to those that control mitosis.
In a healthy organism, cellular proliferation of tissues is normally balanced by cell death, which occurs by programmed apoptosis. Apoptosis is induced via the stimulation of several different cell surface receptors in association with activation of caspases (cysteinyl aspartate-specific proteases). Caspases can be activated by two main pathways: the death receptor pathway and the mitochondrial pathway.
• apoptosis : checkpoints : cyclins ~ cyclin-dependent kinases & cyclin-dependent kinases : G1 : G0 : S : G2 : M : mitosis :
Tables Apoptosis vs Necrosis Apoptosis Regulatory Proteins Sequences Cell signaling Phosphate-handling enzymes Malignant Transformation Oncogenes Proto-oncogenes
DNA replication : meiosis : mitosis : mitotic spindle : replication • A • adhesion • C • CDKs . cell membranes • cellular adhesion molecules • cellular signal transduction • centrioles • chemotaxis • chloroplast • cilia & flagella • communication • concentration gradients . cyclin-dependent kinases • cytokine receptors • cytoplasm • cytoskeleton • E • energy transducers • endoplasmic reticulum • endosomes • exosome • F • flagella & cilia • G • Golgi apparatus • GPCRs • H • hormones • I • ion channels • L • lysosome • M • meiosis • microtubules • mitosis . prophase . anaphase . metaphase . telophase • mitochondrion • N • Nitric Oxide • neurotransmission • neuronal interconnections • nuclear membrane • nuclear pore • P • pinocytosis • proteasome • protein degradation • pumps • R • receptor proteins • receptor-mediated endocytosis • S • second messengers • signaling gradients • signal transduction • spindle • structure • T • transport • two-component systems • U • ubiquitin • V • vacuole • vesicle •
Human Cell Cycle . The Cell Cycle & Mitosis Tutorial . game . Mitosis: An Interactive Animation . Mitosis interactive Java tutorial .
a) Binary fission in prokaryotes and plastids derived from prokaryotes (mitochondria , chloroplasts). In binary fission, which is a form of asexual reproduction, a cell divides transversely into two daughter cells after replication.
b) Budding (yeast, spores, Hydra) is a form of asexual reproduction in which a new organism buds from a cell protuberance in a form of unequal fission.
c) Mitotic division of somatic cells in which DNA is duplicated prior to fission into cells with an equivalent chromosomal complement to that of the parent cells.
c) Meiotic division of germ cells (sexual reproduction) in which fission generates daughter cells with half the chromosomal complement of the parent cells.
 Control of the Cell Cycle (animation):
Control of the Cell Cycle (animation):Right - Cell Cycle - click to enlarge image. Phases G1 and G0, S, G2, M. Checkpoints - purple arrows at G1-S transition, during S phase, G2-M transition, anaphase of mitosis. CDKs (yellow) are modulated by association with a series of fluctuating cyclins (d, e, a, b.) The cycle is divided into non-mitotic interphase (beige) with G1 or G0, S, and G2 phases followed by mitosis (pink).
Eukaryotic cells alternate genome doubling (S-phase, synthesis) with genome splitting (mitosis, M-phase) to generate daughter cells with an identical chromosomal complement. The cell cycle consists of a signal-controlled sequence of physiological states G1 → S → G2 → M, where non-mitotic phases are termed 'interphase' (beige) and G represents 'gap'. Quiescent cells are said to be in phase G0, in which they are not participating in the cell cycle, but are metabolically active.
In a normal resting cell, intracellular signaling proteins and genes remain inactive unless activated by extracellular growth factors. When the normal resting cell is stimulated by an extracellular growth factor, signaling proteins and genes are activated and the cell proliferates.
More than one hundred genes are specifically involved in cell cycle control, these are the so called CDC-genes (cell division cycle genes). One of these genes, designated CDC28 in Saccharomyces cerevisiae or CDK1 (cyclin dependent kinase 1) in humans, controls the first step in the progression through the G1-phase of the cell cycle, and is therefore also called "start". This gene encodes a protein member of the cyclin dependent kinase family (CDKs). Half a dozen different CDK molecules have been found in humans. Cyclins are proteins formed and degraded during each cell cycle. Cyclins are so-named because their levels vary periodically during the cell cycle. Cyclins bind to the CDK molecules, thereby regulating the CDK activity and selecting the proteins to be phosphorylated. Periodic protein degradation is an important general control mechanism of the cell cycle.
Cyclins were conserved during evolution. Around ten different cyclins have been found in humans. The levels of CDK-molecules are constant during the cell cycle, but their activities vary because of the regulatory function of the cyclins. CDK and cyclin together drive the cell from one cell cycle phase to the next. Levels of cyclins D (G1), E and A (S), and B and A (mitotic) fluctuate during the cell cycle (d,e,a,b) , and binding of appropriate cyclins to the cyclin-dependent kinases (CDKs) stimulating phosphorylation and activation.
The cell cycle is a highly coordinated process that is controlled at multiple checkpoints (purple arrows) along the pathway – the G1/S and G2/M transitions of interphase as well as anaphase of mitosis. These checkpoints are critical in preserving the fidelity of the genome. Alterations in the genes that control checkpoint processes are linked to a number of human malignancies including colon, breast, lung, kidney, brain and skin cancers.
During resting conditions, Rb sequesters the E2F transcription factor within the cytosol. Phosphorylation of Rb enables dissociation of the Rb-E2F complex, which allows E2F to translocate to the nucleus, where E2F binds to the promoter region of early genes that prime the cell to progress into the S-phase of the cycle. These early genes include c-myc, cyclin A/E, and CDK1(cdc2).
Progression through the cell cycle begins (G1 → S) with the commitment to undergo cellular division, which is initiated through the activation of the cyclin D-CDK4/6 complex and the subsequent phosphorylation of the Rb-E2F complex. Activity of the cyclin D-CDK4/6 complex is regulated through binding to its respective cyclin (4/6) counterpart, phosphorylation and dephosphorylation events, and the expression of a number of CDK inhibitors (CDKI’s), which . include p15 INK4, p16 INK4, p21 Cip-1 and p27 Kip-1. Progression through G1 is further facilitated by the cyclin E-CDK2 complex.
G1 → S
Rising level of G1-cyclins (D) bind to a G1 Cdk (Cdk 4), signaling the cell to prepare the chromosomes for replication.
DNA-damage checkpoints monitor DNA damage before the cell enters S phase (G1 checkpoint),
during S phase, and after DNA replication (G2 checkpoint). Increased levels of CDK-molecules and cyclins are sometimes found in human cancers. CDK-molecules and cyclins collaborate with the products of tumor suppressor genes, such as TP53 and Rb, during the cell cycle. The p53 protein senses DNA damage and can halt progression of the cell cycle in G1. Both copies of the p53 gene must be mutated for cycle arrest to fail, so mutations in p53 are recessive and p53 qualifies as a tumor suppressor gene. The protein generated by the p53 gene acts as a signal for apoptotic cell death when DNA damage is too extensive for repair mechanisms.
S → G2
Rising level of S-phase promoting factor (SPF) — which includes cyclin A bound to Cdk2 — enter the nucleus and prepare the cell to duplicate its DNA and its centrosomes. As DNA replication continues, cyclin E is destroyed, and the level of mitotic cyclins begins to rise (in G2).
In the S-phase of the cycle, the DNA is unwound and replicated. If DNA is damaged, the fate of the cell is determined by the extent of damage. If the damage is repairable, progression through the cycle will be halted in a p53-dependent matter through p21 Cip-1, allowing sufficient time to repair the damage.
Cells monitor the presence of the Okazaki fragments on the lagging strand during DNA replication, and cells do not proceed in the cell cycle until these have disappeared.
Entry into the M-phase of the cell cycle is regulated by the cyclin B-CDK1 complex. CDK1 activity is subject to multiple levels of control in an analogous fashion to the other cyclin-dependent kinases.
G2 → M
M-phase promoting factor (a complex of mitotic cyclins with the M-phase Cdk) initiates metaphase assembly of the mitotic spindle, breakdown of the nuclear envelope, condensation of chromosomes.
Spindle checkpoints monitor any failure of spindle fibers to attach to kinetochores and arrest the cell in metaphase until all the kinetochores are attached correctly (M checkpoint), detect improper alignment of the spindle itself, block cytokinesis, and trigger apoptosis if the damage is irreparable.
M-phase promoting factor activates the anaphase-promoting complex (APC/C, cyclosome) which allows the sister chromatids assembled at the metaphase plate to separate and move to the poles during telophase, completing mitosis. The APC/C destroys cyclin B by attaching it to the protein ubiquitin, thus targetting it for destruction by proteasomes (hydrolysis by proteases). APC/C also turns on synthesis of G1 cyclin for entry into the next turn of the cycle, degrades geminin, a protein that has retained the freshly-synthesized DNA in S phase to prevent if from being re-replicated before mitosis.
This is but one mechanism by which the cell ensures that every portion of its genome is copied once — and only once — during S phase. Some cells deliberately cut the cell cycle short allowing repeated S phases without completing mitosis and/or cytokinesis (endoreplication).
In a cancer cell, mutation in a proto-oncogene that encodes an intracellular signaling protein (normally activated only by extracellular growth factors) creates an oncogene. The oncogene encodes an altered form of the signaling protein that behaves as though activated despite the absence of growth factor binding. That is, the malignant cell has escaped normal gene regulation and cell cycle control mechanisms and exhibits unchecked proliferation.
Tables Regulatory Proteins Sequences Cell signaling Cell Adhesion Molecules Second Messengers Immune Cytokines Malignant Transformation Oncogenes Proto-oncogenes
Meiosis is controlled by similar factors to those that control mitosis.
In a healthy organism, cellular proliferation of tissues is normally balanced by cell death, which occurs by programmed apoptosis. Apoptosis is induced via the stimulation of several different cell surface receptors in association with activation of caspases (cysteinyl aspartate-specific proteases). Caspases can be activated by two main pathways: the death receptor pathway and the mitochondrial pathway.
• apoptosis : checkpoints : cyclins ~ cyclin-dependent kinases & cyclin-dependent kinases : G1 : G0 : S : G2 : M : mitosis :
Tables Apoptosis vs Necrosis Apoptosis Regulatory Proteins Sequences Cell signaling Phosphate-handling enzymes Malignant Transformation Oncogenes Proto-oncogenes
DNA replication : meiosis : mitosis : mitotic spindle : replication • A • adhesion • C • CDKs . cell membranes • cellular adhesion molecules • cellular signal transduction • centrioles • chemotaxis • chloroplast • cilia & flagella • communication • concentration gradients . cyclin-dependent kinases • cytokine receptors • cytoplasm • cytoskeleton • E • energy transducers • endoplasmic reticulum • endosomes • exosome • F • flagella & cilia • G • Golgi apparatus • GPCRs • H • hormones • I • ion channels • L • lysosome • M • meiosis • microtubules • mitosis . prophase . anaphase . metaphase . telophase • mitochondrion • N • Nitric Oxide • neurotransmission • neuronal interconnections • nuclear membrane • nuclear pore • P • pinocytosis • proteasome • protein degradation • pumps • R • receptor proteins • receptor-mediated endocytosis • S • second messengers • signaling gradients • signal transduction • spindle • structure • T • transport • two-component systems • U • ubiquitin • V • vacuole • vesicle •
Human Cell Cycle . The Cell Cycle & Mitosis Tutorial . game . Mitosis: An Interactive Animation . Mitosis interactive Java tutorial .










































0 Comments:
Post a Comment
<< Home